Scientists won’t know definitively if any of the vaccines work until next year
Lead researcher overseeing a clinical vaccine trial for coronavirus has said that scientists won't know definitively if any vaccine works to prevent COVID-19.
- World News
- 2 min read
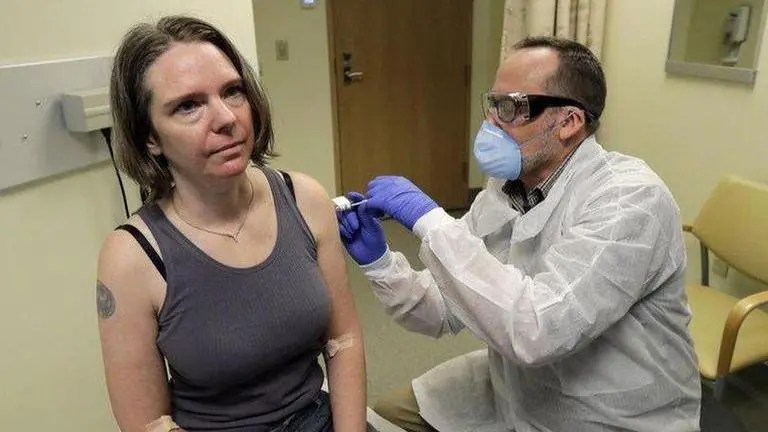
The lead researcher overseeing a clinical vaccine trial for coronavirus has said that scientists won't know definitively if any vaccine works to prevent COVID-19 at least till next year. This comes as the coronavirus infection, which emerged in the Chinese city of Wuhan has now spiralled to infect 3,822,295 people across the world with the majority in the US. Out of the total infected, 1,303,793 of have recovered and 265,116 died.
Speaking to an American media outlet, Dr Mark Mulligan said that a one-year time frame is ‘blazing time frame’ for vaccine development. He added that what they were doing in the month was a work to be done in years. Mulligan is the director of Vaccine centre at NYU’s Langone Health. NYU Grossman School of Medicine and the University of Maryland were first centres to enrol patients in a vaccine trial for COVID-19.
Advertisement
Mulligan added that the first phase of the trial will determine if it is safe adding that it would take nearly three to four weeks. He further said that safety was the first question following which they’ll test if it was tolerated well by the body and if it produces antibody responses that might work even after those three-four months.
Human trials at Oxford
On the other hand, researchers at the University of Oxford are confident of making available coronavirus vaccine by September. Sarah Gilbert, the lead researcher of the vaccine development programme, reportedly told a virtual conference on April 17 that her team is confident about the efficacy of ChAdOx1 vaccine. Meanwhile, Norwegian diplomat Erik Solheim crossed fingers in hope at a report which stated that researchers at Oxford University are working on a vaccine for Coronavirus and it could be ready as early as September - far before the 2021 minimum timeframe earlier suggested.
Advertisement
Let’s cross our fingers!
— Erik Solheim (@ErikSolheim) April 12, 2020
A coronavirus vaccine could be ready for the public by September. Human trials are scheduled to start in a few weeks
pic.twitter.com/Mz3cOvtvq5
(Image credits: AP)
