Alzheimer’s Treatment Drug Launched In India At Nearly ₹1 Lakh Per Dose, Here's All You Need To Know
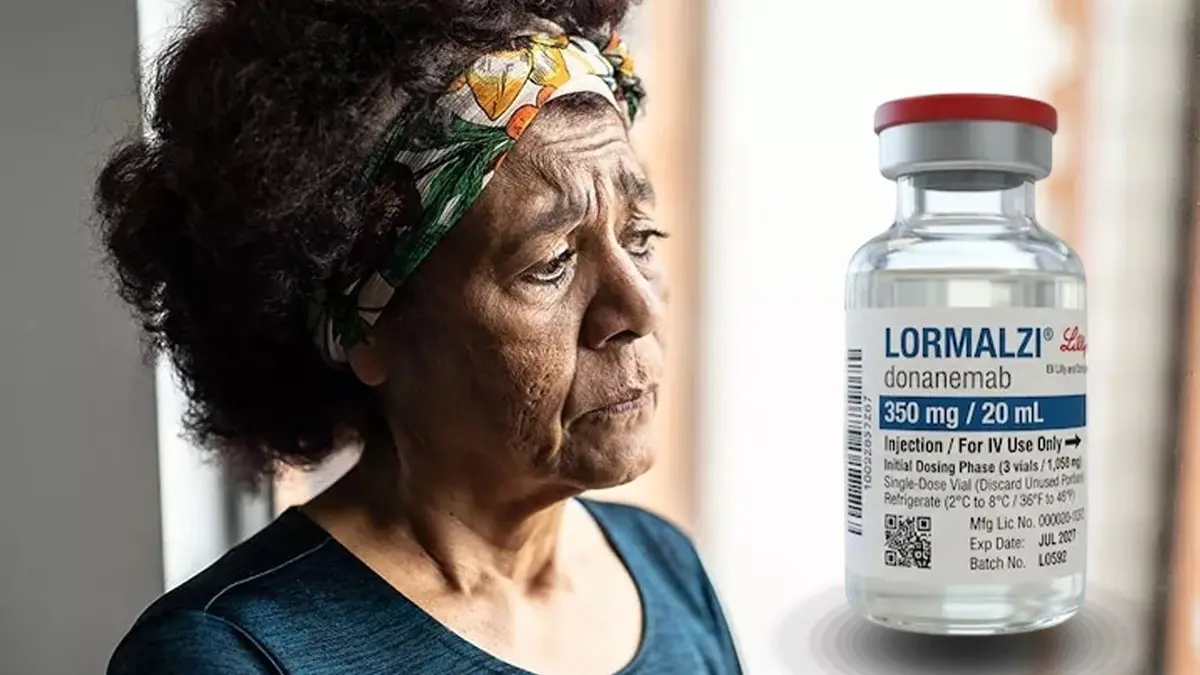
Priced at Rs 91,688 for a 350 mg vial, the prescription-only intravenous therapy is to be administered once a month for up to a period of 18 months.
- Health News
- 3 min read
Alzheimer's disease is the most common cause of dementia. Alzheimer's is the biological process that begins with the appearance of a buildup of proteins in the form of amyloid plaques and neurofibrillary tangles in the brain. This causes brain cells to die over time and the brain to shrink. Early symptoms of Alzheimer's include forgetting recent events or conversations. Over time, Alzheimer's disease leads to serious memory loss and affects a person's ability to do everyday tasks. Old people are commonly diagnosed with Alzheimer's. In advanced stages, loss of brain function can cause dehydration, poor nutrition or infection. These complications may result in death.
Also read: Can Summer Heat Affect Your Period?
At least 10 million new cases of Alzheimer's are expected to be reported annually, and the number of affected patients may rise up to 139 million globally by 2050. According to estimates cited by Lilly, dementia affects approximately 8.8 million people in India, with Alzheimer's disease accounting for the majority of cases. The number of patients is projected to nearly double by 2036 here. Given the global health scare this disease poses to be, India has received a new treatment option for the early onset of Alzheimer's. Eli Lilly and Company (India) launched Lormalzi (donanemab), a once-monthly amyloid plaque-targeting therapy approved for patients with mild cognitive impairment or mild dementia due to Alzheimer's disease.

Advertisement
Priced at Rs 91,688 for a 350 mg vial, the prescription-only intravenous therapy has received marketing authorisation from the Central Drugs Standard Control Organisation (CDSCO). Lilly says the drug is the first and only once-monthly amyloid plaque-targeting therapy currently available in India, designed specifically for patients in the early symptomatic stages of Alzheimer's disease.
The therapy is approved for:
Advertisement
– Patients with mild cognitive impairment due to Alzheimer's disease
– Those in the mild dementia stage, provided amyloid pathology is confirmed.

Lormalzi is administered as a once-monthly intravenous infusion. Unlike conventional Alzheimer's medicines that mainly address symptoms temporarily, donanemab is designed to target the underlying disease biology by reducing amyloid plaque accumulation. Amyloid plaques are abnormal protein deposits that accumulate between nerve cells in the brain and are believed to contribute to memory loss, impaired thinking and cognitive decline associated with Alzheimer's disease. Treatment in India is expected to continue for 18 months (76 weeks) in most patients with this drug.
