US FDA issues warning letters to two groups for selling fake COVID-19 vaccines
US FDA and the Federal Trade Commission reportedly issued warning letters to two groups for selling fraudulent products related to COVID-19.
- World News
- 3 min read
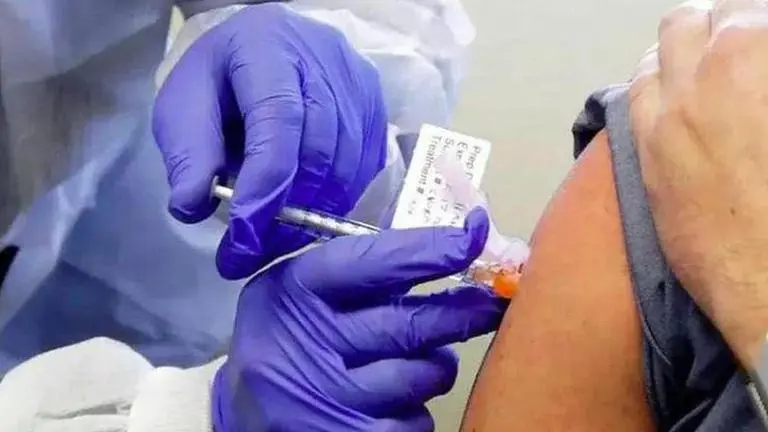
The United States Food and Drug Administration (FDA) and the Federal Trade Commission reportedly issued warning letters to two groups for selling fraudulent products related to COVID-19. According to an international media outlet, Apollo Holding LLC and North Coast Biologics were selling fake vaccines cannabidiol (CBD) oil for coronavirus.
Apollo Holding was reportedly offering ‘NoronaPak’ products which include CBD, derived from the cannabis plant. According to the reportedly warning letter, the products were ‘intended to mitigate, prevent, treat, diagnose, or cure COVID-19 n people’. The FDA said that the dugs were unproven and sold in violation of federal law. Apollo Holding reportedly also made claims such as ‘Fight off coronavirus with NoronaPak’.
According to the letter issued to North Coast Biologics, the company was allegedly offering ‘nCoV19 spike protein vaccine’. The FDA in the letter reportedly also pointed out that a person involved with North Coast Biologics also bragged about the supposed vaccine of social media platform. The employee reportedly claimed that the company vaccinated 12 people in West Seattle and they were also going to vaccinate 12 more in Burien.
Advertisement
The letter further noted that the social media post also read that NCB’s COVID-19 spike protein vaccine is being made available to those who are either at risk or for anyone who simply needs some reassurance. After the warning from FDA and FTC, the post has, however, been edited. According to FDA the edited version now reads that the vaccine is no longer available due to ‘cease and desist’ letter. Although FDA and FTC also reportedly said that misleading claims still remained online at the time of the warning.
Advertisement
First at-home test
Meanwhile, earlier this month, FDA authorised the first diagnostic test with the option of using home-collected saliva samples for COVID-19. According to the press note on FDA website, the association issued an emergency use authorisation to Rutgers Clinical Genomics Laboratory for their COVID-19 laboratory-developed test as well. With the test, people can collect their own saliva at home and send the sample to a lab for results.
The FDA press note also mentioned that RUCDR’s test is currently the only authorised COVID-19 diagnostic test that uses saliva samples to test for the deadly virus. The association also noted that the test is ‘not a general authorisation’ for at-home collection of a patient sample using other collection methods, saliva collection devices, or tests, or for tests fully conducted at home.
