Published 11:48 IST, February 9th 2022
FabiSpray: Glenmark launches COVID nasal spray in India for adults; check details
Glenmark Pharmaceuticals Limited (Glenmark) has launched Nitric Oxide Nasal Spray, under the brand name FabiSpray for the treatment of adult COVID patients.
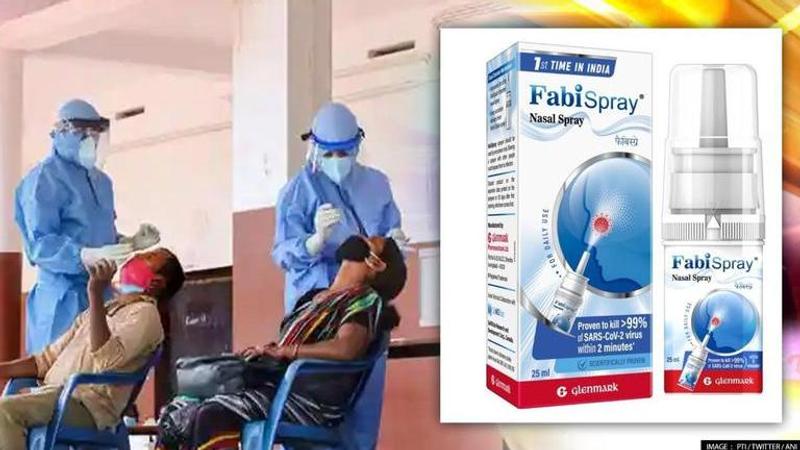
Image: PTI/Twitter_@ANI | Image:
self
- Listen to this article
- 3 min read
Advertisement
11:48 IST, February 9th 2022
